
After sharing 2 valence electrons with hydrogen atoms to form single covalent bond, each carbon atom now needs two more valence electrons and share two valence electrons with each other to form double covalent bonds between two carbon atoms. Hydrogen atom needs one more electron to be stable and carbon atoms will share 2 valence electrons with 2 hydrogen atoms. Each atom now has eight valence electrons and is stable.Ī carbon atom has four valence electrons, it needs four more electrons to be stable. Two nitrogen atoms share three electrons each, forming triple covalent bonds in the molecule with the formula N 2. 
Describe the electron arrangement in more complex covalent molecules such as N 2, C 2H 4, CH 3OH and CO 2Ī nitrogen atom has five valence electrons, it needs three more electrons to be stable.Usually do not conduct electricity as there are no charged particles, no mobile ions or electrons Usually insoluble in water, soluble in organic solventsĬonduct electricity in molten and aqueous state due to mobile ions, do not conduct in solid state Usually soluble in water, insoluble in organic solvents Small amount of energy is needed to overcome these forces. Low melting and boiling points due to Simple molecular structure with weak intermolecular forces. Large amount of energy is needed to break these bonds. High melting and boiling points due Giant ionic lattice structure with strong ionic bonds. 
Describe the differences in volatility, solubility and electrical conductivity between ionic and covalent compounds.nitrogen and hydrogen have a stable electronic configuration of 2.8 and 2 respectively. Each hydrogen atom shares one electron with nitrogen atom. Nitrogen atom shares 3 valence electrons with three hydrogen atoms. Nitrogen atom needs 3 electron more to be stable. Carbon and hydrogen have a stable electronic configuration of 2.8 and 2 respectively. Each hydrogen atom shares one electron with carbon atom. The carbon atom shares 4 valence electrons with four hydrogen atoms. Oxygen and hydrogen have a stable electronic configuration of 2.8 and 2 respectively.Ĭarbon atom needs 4 more electrons to be stable. Each hydrogen atom shares one electron with oxygen atom. The oxygen atom shares 2 valence electrons with the two hydrogen atoms. Oxygen atom needs two more electrons and hydrogen atom need one more electron to be stable. Chlorine and hydrogen then will have a stable electronic configuration of 2.8.8 and 2 respectively. 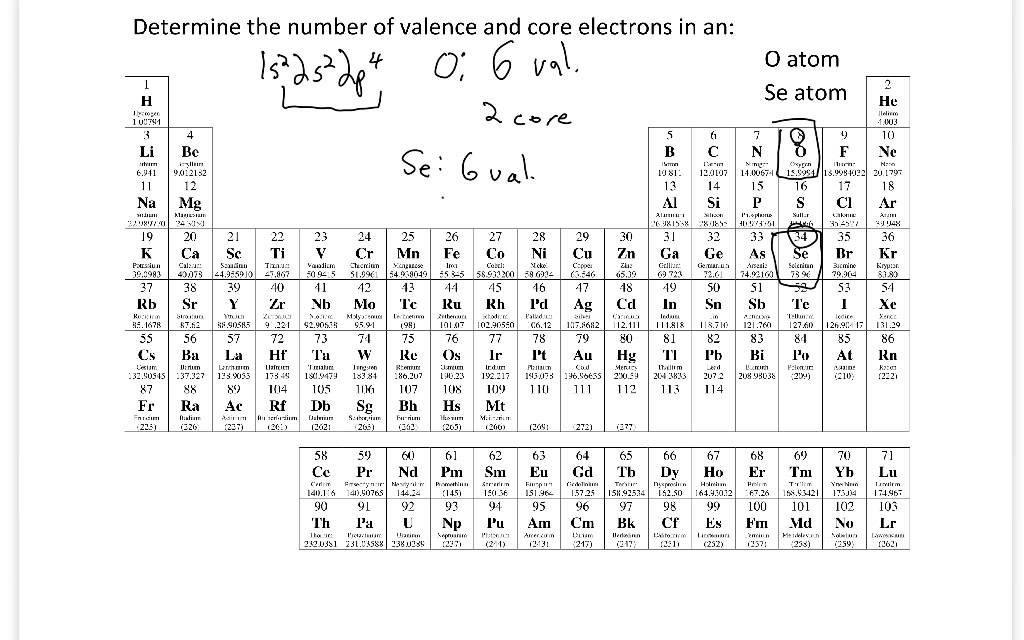
The chlorine atom and hydrogen atom share one electron with each other. Two chlorine atoms can share one electron with each other to achieve stable electronic configuration for the valence electron shell.Įach chlorine and hydrogen atom need one more electron to be stable. Each chlorine atom needs one more electron to be stable. Two hydrogen atoms can share one electron with each other to achieve stable electronic configuration for the first electron shell.Ī chlorine atom has 7 valence electrons. Each hydrogen atom needs one more electron to be stable.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
